| Congenital | Post-inflammatory | Infective | Neoplastic | Other |
|---|---|---|---|---|
|
|
|
|
|
Vitiligo’s effect is not just skin deep

Vitiligo is a chronic autoimmune condition characterised by the complete loss of pigmentation in discrete patches of skin.2,3 Affecting around 100 million people globally, it has a prevalence of around 0.4–1.6%.4–7 Non-segmental vitiligo accounts for 84–95% of vitiligo cases.8
Beyond its visible effects, vitiligo places a substantial psychological burden on patients that often impacts their mental health.4,9–11 Additionally, it is associated with various autoimmune comorbidities.12–14
As a first point of contact, GPs are ideally placed to make an initial diagnosis of vitiligo, provide first-line treatment and supportive care, as well as assess the need for referral to dermatology.15,16 There is currently an unmet need for treatment options for patients with vitiligo.16 This page aims to support GPs in managing their patients with vitiligo and raise awareness of Opzelura® topical cream – a NICE recommended, licensed treatment for non-segmental vitiligo available in secondary care to aid repigmentation of the skin.1,17,18
Diagnosis of vitiligo
A clinical diagnosis of vitiligo is typically based on an assessment including a full medical history and an examination of the lesions to determine the morphology and distribution.3,16 Investigations should be considered to check for associated autoimmune diseases, and a differential diagnosis should be ruled out.3,16,19
There are several different types of vitiligo with different classifications and nomenclature, including:2
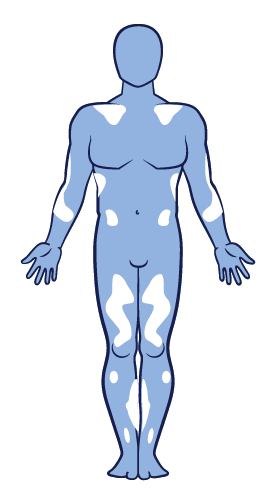 Non-segmental vitiligo2–
Non-segmental vitiligo2–the most common
type of vitiligo20
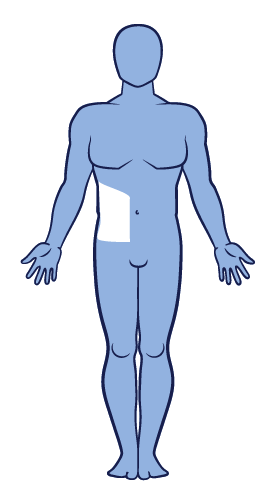 Segmental vitiligo2
Segmental vitiligo2 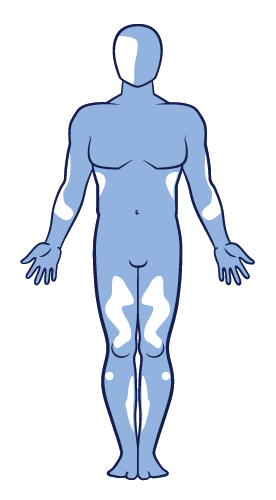 Mixed vitiligo2
Mixed vitiligo2 Learn more about each type of vitiligo.
Opzelura® is not indicated for the treatment of segmental vitiligo or mixed vitiligo.1
History and examination
For a patient with suspected vitiligo, take a full medical history, examine the patient and document the following:3,16,21,22
- Age of onset
- Any known triggers, such as occupational exposure to chemicals
- Site and type of vitiligo (segmental or non-segmental)
- Extent of lesions (affected BSA) and appearance
- Disease stability (check for the presence of Koebner’s phenomenon)
- Fitzpatrick skin type
- Quality of life
- Psychosocial and psychological impact
- Type or duration of any previous treatments to manage the condition
- Any known risk factors, such as a family history of vitiligo or a personal or family history of thyroid dysfunction or other autoimmune disease
If available, consider examining the lesions with a Wood's lamp (UV lamp) to help to confirm a diagnosis of vitiligo:23
- Depigmented areas fluoresce a bright white colour
- Margins are sharply delineated
- Reveals the actual size of lesions
Investigations
Screen for antithyroid antibodies and thyroid function to identify patients at high risk of developing autoimmune thyroid disease.16 Consider blood tests for other comorbid autoimmune diseases if suggested by the patient's history and/or other test results.21
Differential diagnosis
Consider a differential diagnosis, as there are many conditions that can mimic vitiligo, presenting with areas of depigmentation:3,15,19
Scroll right if necessary

Please note that Opzelura® is not licensed for use in any of the conditions listed in the table above.1
Management in primary care
The management of non-segmental vitiligo requires consideration of skin phenotype, disease stability, lesion extent and distribution, as well as patient age, comorbidities and preferences:24
- Provide your patient with information about their vitiligo, discuss available treatment options and advise them on sources of support16,24
- Advise your patient on the importance of sun protection and vitamin D supplementation16
- Consider a skin camouflage consultation for patients who would like to explore this option:16,24
- Assess and monitor the quality of life and level of psychological distress associated with living with vitiligo:24
- Manage any associated psychosocial comorbidities24
- Reassess the use of topical treatments every 3–6 months to assess response and to monitor for adverse effects16,24
Treatment options for non-segmental vitiligo
Consider the following first-line treatment options for your patients:16
- Potent or very potent TCS
- TCI (such as tacrolimus) for facial vitiligo as an alternative to potent or very potent TCS
- An intermittent regimen of once-daily potent or very potent TCS with or without TCI, especially for patients with vitiligo especially in areas with thinner skin (such as the periocular region, genital area and skin flexures)
Advise patients that an approved treatment is available in secondary care to aid the repigmentation of the skin.1,17
The importance of referral to secondary care
Consider referral for patients regardless of Fitzpatrick skin type
or duration of
vitiligo1,25
It is important to have conversations with your vitiligo patients to discuss current available vitiligo treatments, and the psychosocial impact that vitiligo may be having on their lives.
Effective vitiligo management often extends beyond medication alone. Prioritising shared decision-making and meaningful patient conversations is essential; this personalised approach ensures that treatment goals align with the vitiligo patients' preferences, lifestyles, and expectations.25
Patients should be fully informed of their condition, available treatment options and the benefits of specialist care and support services. Referral should only be made after obtaining agreement from your patient as part of a shared care decision.25

Consider referral for treatment with Opzelura® – the first and only approved therapy for the treatment of non-segmental vitiligo*1,26
Opzelura®, a topical JAK 1/2 inhibitor, is the only approved treatment to aid quality repigmentation of non-segmental vitiligo and has been approved for use in Europe and the UK.*1,26
Opzelura® works by inhibiting JAK-STAT signalling to address the three vitiligo treatment goals:1,27
- Halting depigmentation – by selectively binding to JAK1 and JAK21
- Inducing repigmentation – melanocytes migrate from hair follicles and lesion edges17
- Preventing relapses – selective JAK1 binding inhibiting signal transmission by IL-1528,29
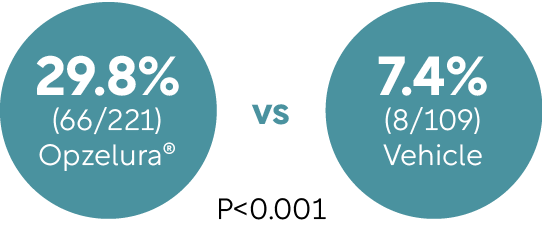
In two pivotal randomised studies, TRuE-V1 and TRuE-V2, Opzelura® demonstrated significant repigmentation over time and was generally well tolerated with few sides effects and a low treatment-related discontinuation rate of <1%.30
After 52 weeks, around 30% of patients (n=173) achieved ≥90% facial repigmentation (F-VASI) with Opzelura® and one in two patients (91/173) achieved a 75% improvement in facial pigmentation.30
Opzelura® is now recommended by NICE for your patients with non segmental vitiligo18
NICE recommends Opzelura® as an option to treat non-segmental vitiligo with facial involvement in people 12 years and over only if:18
- Topical first-line treatments have not worked or are not suitable, and
- The company provides it according to the commercial arrangement
For eligible patients whose primary goal of vitiligo treatment is repigmentation, offer a referral to dermatology so they can discuss treatment with Opzelura®.1,17,18,25
When to refer to dermatology
Refer to dermatology in agreement with your patient in the case of:16,24,25
- Rapid progression
- Diagnostic uncertainty
- Segmental vitiligo
- Widespread vitiligo affecting large areas of the body (>10% BSA)
- Significant psychosocial impact
- Inadequate response to topical treatments
- Contraindications to or adverse events from topical treatments
Other reasons for referral
Offer referral to:16,24,25
- A local skin camouflage service if your patient wishes to camouflage their lesions
- Psychological services for group or/and individual CBT to patients with moderate-to-severe psychological distress
Refer eligible patients for treatment with Opzelura®
Opzelura® is now recommended by NICE and is available on the NHS in England and Wales for your patients with non-segmental vitiligo.18
For patients with suspected vitiligo, refer promptly to secondary care or a GPwER to support timely access to treatment options.
If you have any questions about Opzelura® get in touch via the link below.
Get in touchOpzelura® is contraindicated during pregnancy and breastfeeding.1
*Opzelura® is indicated for the treatment of non-segmental vitiligo with facial involvement in adults and adolescents from 12 years of age.1
Abbreviations
BSA, body surface area; CBT, cognitive behavioural therapy; F-VASI, Facial Vitiligo Area Scoring Index; GPwER, GP with Extended Role; IL-15, interleukin-15; JAK, Janus kinase; STAT, signal transducer and activator of transcription; TCI, topical calcineurin inhibitor; TCS, topical corticosteroid; UV; ultraviolet.
References
- Opzelura® (ruxolitinib cream) Summary of Product Characteristics. Incyte Biosciences.
- Bergqvist C, et al. Dermatology. 2020;236:571–592.
- Primary Care Dermatology Society. Vitiligo. Available at: https://www.pcds.org.uk/clinical-guidance/vitiligo (Accessed April 2026).
- Bergqvist C, et al. J Dermatol. 2021;48(3):252–270.
- VR Foundation. About Vitiligo. Available at: https://vrfoundation.org/about_vitiligo_posts/how-many-people-have-vitiligo (Accessed April 2026).
- Bibeau K, et al. J Eur Acad Dermatol Venereol. 2022;36:1831–1844.
- Krüger C, et al. Int J Dermatol. 2012;51:1206–1212.
- Picardo M, et al. Nat Rev Dis Primers. 2015;1:15011.
- Krüger C, et al. Acta Derm Venereol. 2015;95(5):553–558.
- Bibeau K, et al. JAMA Dermatol. 2023;159(10):1124–1128.
- Phan K, et al. Pigment Int. 2022;9(2):127–130.
- Hadi A, et al. J Am Acad Dermatol. 2020;82(3):628–633.
- Gill L, et al. J Am Acad Dermatol. 2016;74(2):295–302.
- Korde S, et al. MVP J Med Sci. 2018;5(2):125–133.
- Vitiligo Society. Diagnosis and how your GP can help. Available at: https://vitiligosociety.org/how-your-gp-can-help/ (Accessed April 2026).
- Eleftheriadou V, et al. Br J Dermatol. 2022;186:18–29.
- Birlea SA, et al. Dermatol Clin. 2017;35(2):205–218.
- NICE. Ruxolitinib cream for treating non-segmental vitiligo in people 12 years and over. Technology appraisal guideline GID-TA11949. February 2026. Available at: https://www.nice.org.uk/guidance/gid-ta11949/documents/674 (Accessed April 2026).
- CKS. Vitiligo. Differential diagnosis. Available at: https://cks.nice.org.uk/topics/vitiligo/diagnosis/differential-diagnosis/ (Accessed April 2026).
- Ezzedine K, et al. Lancet. 2015;386:74–84.
- CKS. Vitiligo. Assessment. Available at: https://cks.nice.org.uk/topics/vitiligo/diagnosis/assessment/ (Accessed April 2026).
- CKS. Vitiligo. Diagnosis. Available at: https://cks.nice.org.uk/topics/vitiligo/diagnosis/diagnosis/ (Accessed April 2026).
- Dyer JM, Foy VM. J Clin Aesthet Dermatol. 2022;15(6):25–30.
- CKS. Vitiligo. Management. Available at: https://cks.nice.org.uk/topics/vitiligo/management/ (Accessed April 2026).
- Anbar T, et al. Exp Dermatol. 2014;23(4):219–223.
- Hospital Pharmacy Europe. Ruxolitinib cream gains MHRA approval for non-segmental vitiligo. Available at: https://hospitalpharmacyeurope.com/clinical-zones/dermatology/ruxolitinib-cream-gains-mhra-approval-for-non-segmental-vitiligo/ (Accessed April 2026).
- Passeron T. Dermatol Clin. 2017;35(2):163–170.
- Chen X, et al. Free Radic Biol Med. 2019;139:80–91.
- Nolz JC, Richer MJ. Mol Immunol. 2020;117:180–188.
- Rosmarin D, et al. N Engl J Med. 2022:387(16):1445–1455.
UNITED KINGDOM
Adverse events should be reported.
Reporting forms and information can be found at:
www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in the Google Play or Apple App Store.
Adverse events should also be reported to Incyte by calling 00-800-0002-7423.
REPUBLIC OF IRELAND
Adverse events should be reported.
Reporting forms and information can be found at HPRA Pharmacovigilance:
www.hpra.ie. Adverse events should also be reported to Incyte by calling
1800‑456‑748.

